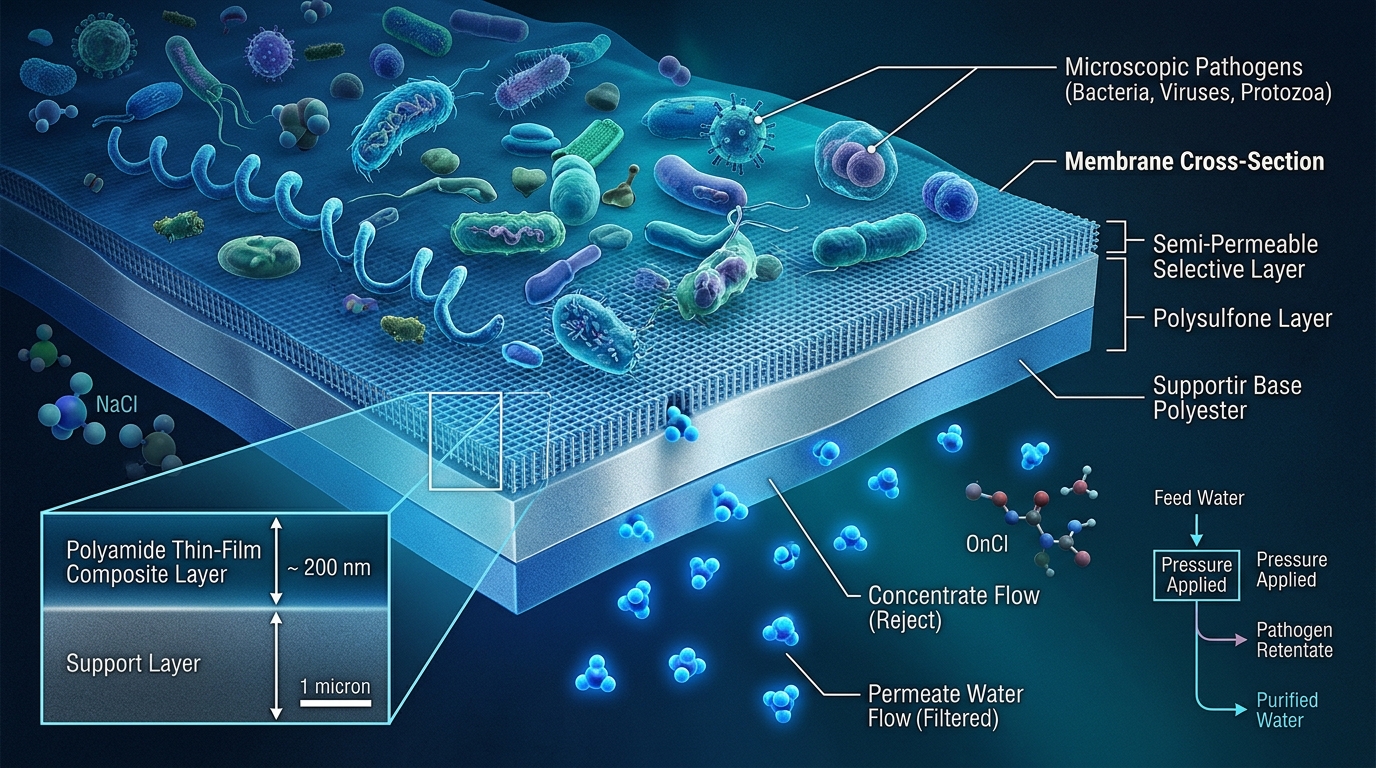
Quick Answer: Norovirus and hepatitis A virus are the leading causes of acute viral gastroenteritis and foodborne/waterborne hepatitis respectively. Detecting these pathogens in surface water at concentrations relevant to public health risk requires highly sensitive molecular detection methods. Research developin. Advanced water treatment technologies including reverse osmosis provide effective solutions for water quality challenges in this area. AMPAC USA’s commercial and industrial systems are engineered to address these specific water treatment needs with certified, documented performance.
Borgmastars, E.; Jazi, M. M.; Persson, S.; Jansson, L.; Radstrom, P.; Simonsson, M.; Hedman, J.; Eriksson, R.
Food and Environmental Virology, 9 (4):395-405; 10.1007/s12560-017-9295-32017
Abstract: Quantitative reverse transcriptase polymerase chain reaction (RT-qPCR) detection of waterborne RNA viruses generally requires concentration of large water volumes due to low virus levels. A common approach is to use dead-end ultrafiltration followed by precipitation with polyethylene glycol. However, this procedure often leads to the co-concentration of PCR inhibitors that impairs the limit of detection and causes false-negative results. Here, we applied the concept of pre-PCR processing to optimize RT-qPCR detection of norovirus genogroup I (GI), genogroup II (GII), and hepatitis A virus (HAV) in challenging water matrices. The RT-qPCR assay was improved by screening for an inhibitor-tolerant master mix and modifying the primers with twisted intercalating nucleic acid molecules. Additionally, a modified protocol based on chaotropic lysis buffer and magnetic silica bead nucleic acid extraction was developed for complex water matrices. A validation of the modified extraction protocol on surface and drinking waters was performed. At least a 26-fold improvement was seen in the most complex surface water studied. The modified protocol resulted in average recoveries of 33, 13, 8, and 4% for mengovirus, norovirus GI, GII, and HAV, respectively. The modified protocol also improved the limit of detection for norovirus GI and HAV. RT-qPCR inhibition with C q shifts of 1.6, 2.8, and 3.5 for norovirus GI, GII, and HAV, respectively, obtained for the standard nucleic acid extraction were completely eliminated by the modified protocol. The standard nucleic acid extraction method worked well on drinking water with no RT-qPCR inhibition observed and average recoveries of 80, 124, 89, and 32% for mengovirus, norovirus GI, GII, and HAV, respectively.
https://link.springer.com/article/10.1007%2Fs12560-017-9295-3
The post Improved Detection of Norovirus and Hepatitis A Virus in Surface Water by Applying Pre-PCR Processing appeared first on Facts About Water.
Source: Water Feed
AMPAC USA engineers custom water purification systems for commercial, industrial, and emergency applications — from 500 GPD to multi-million GPD. Trusted by municipalities, military, and industry worldwide.





![Reverse Osmosis Membrane Lifespan: How Long Do RO Membranes Last? [Maintenance Guide]](https://www.ampac1.com/blog/wp-content/uploads/2026/03/membrane-lifespan-150x150.png)
